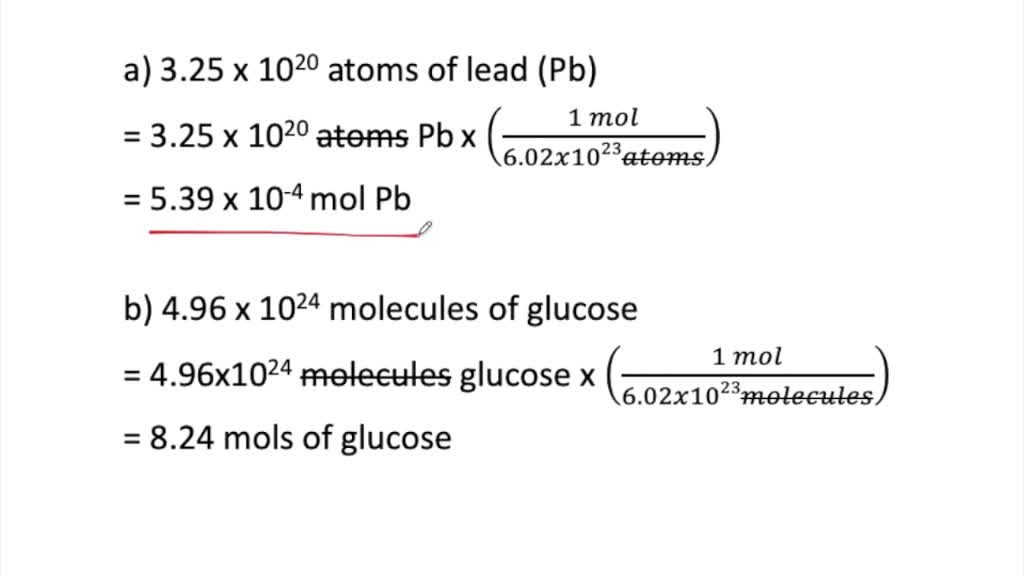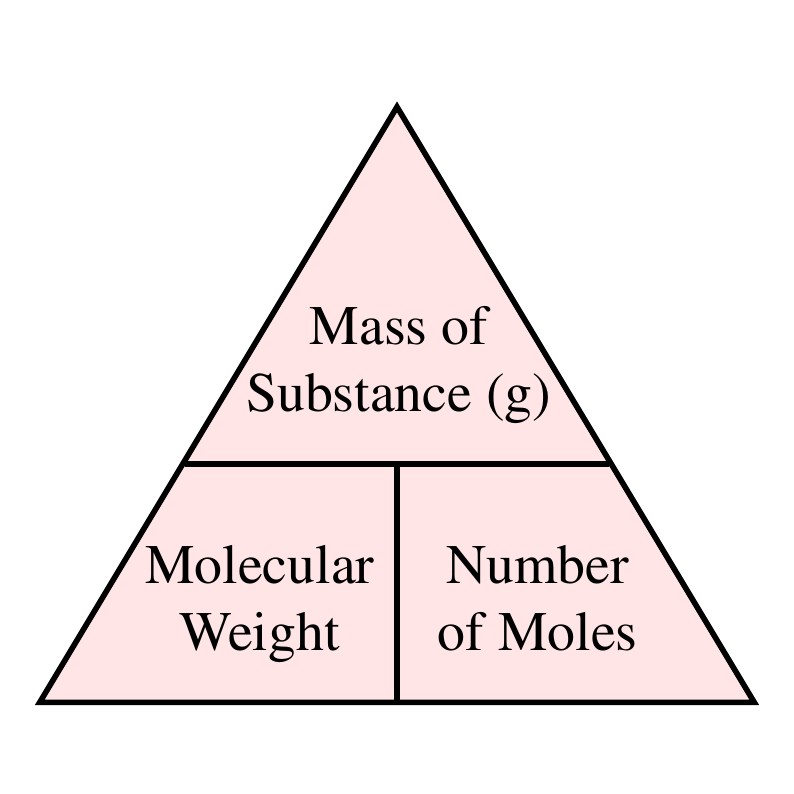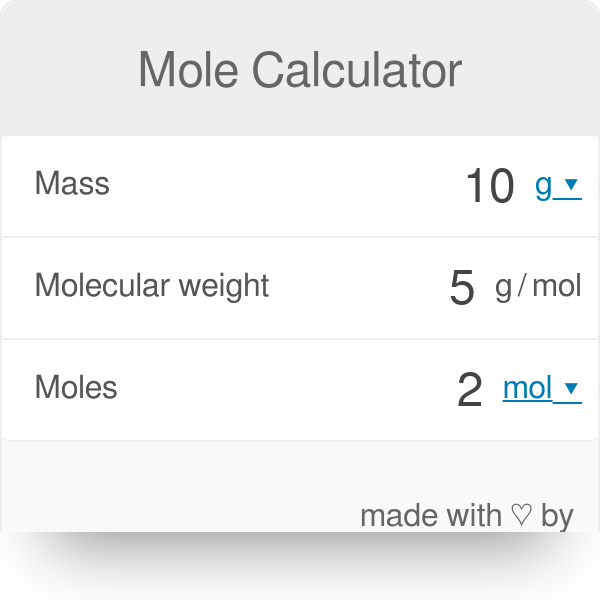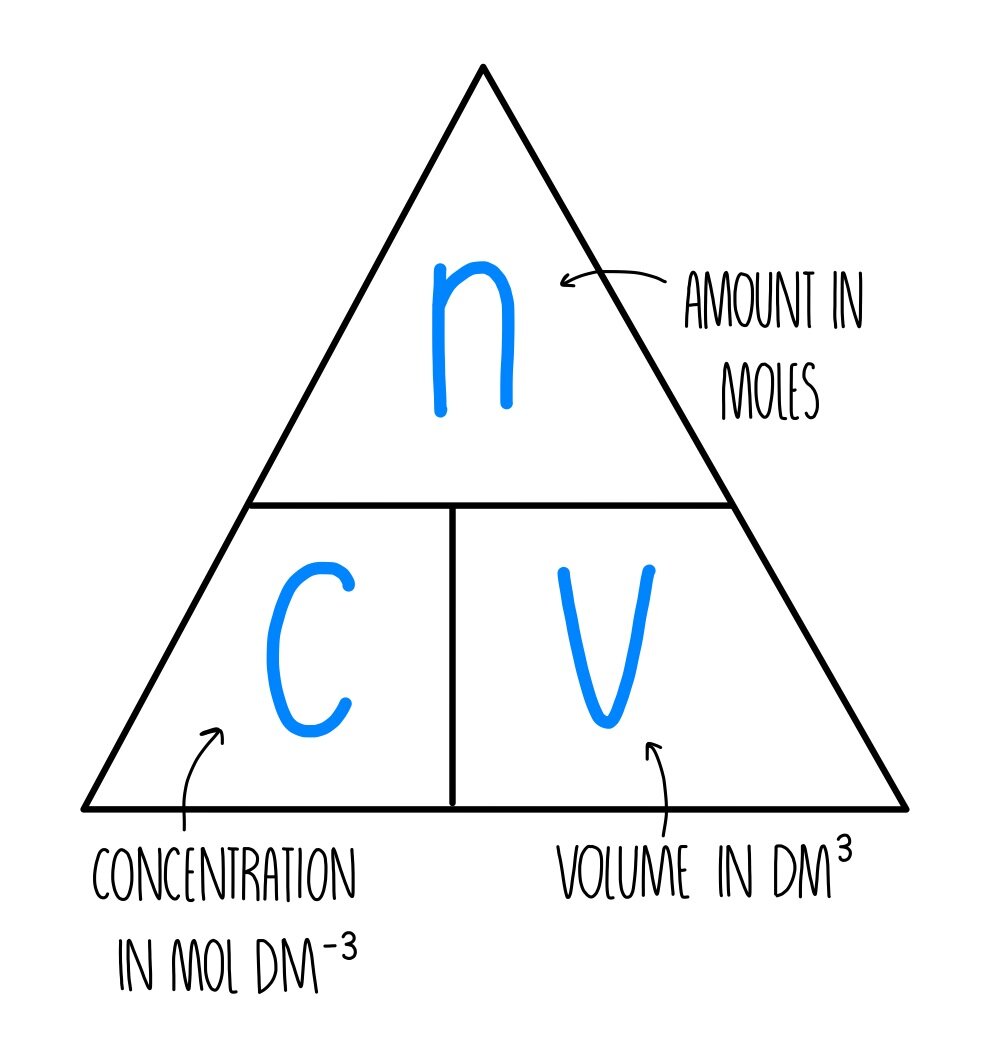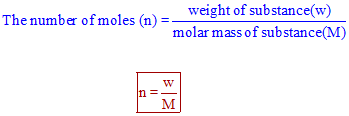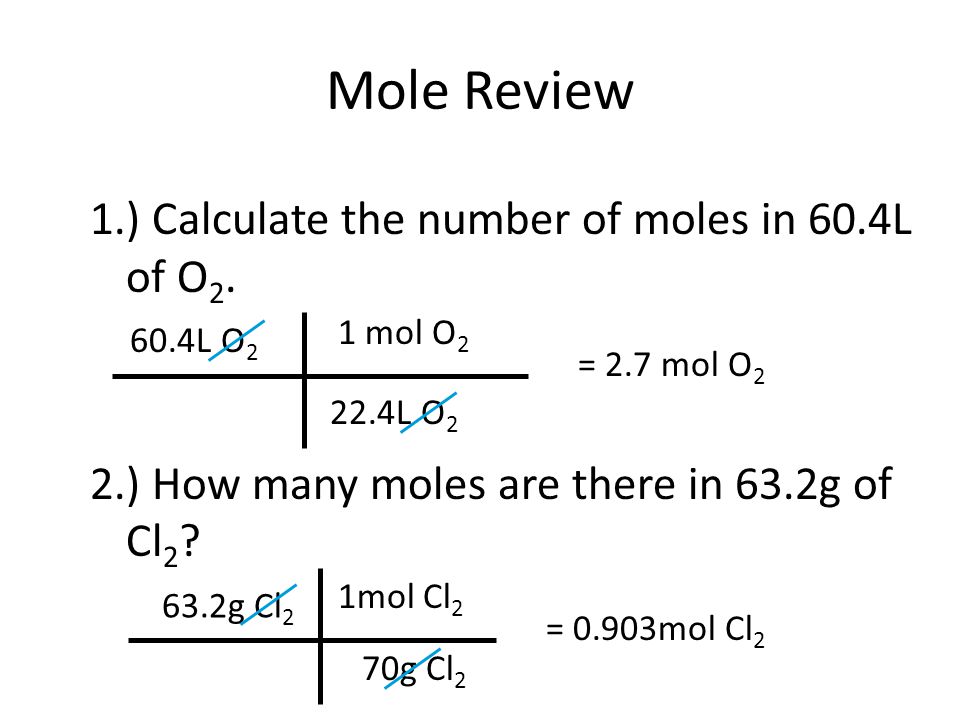
Mole Review 1.) Calculate the number of moles in 60.4L of O2. 2.) How many moles are there in 63.2g of Cl2? 1 mol O2 60.4L O2 = 2.7 mol O2 22.4L

SOLVED: How many moles of HCI are present in the average titre volume? The mole of HCI = 0.001 mole Na2cO3* mole HCI 1 mole Na2co3 0.002 mole HCI Calculate the concentration

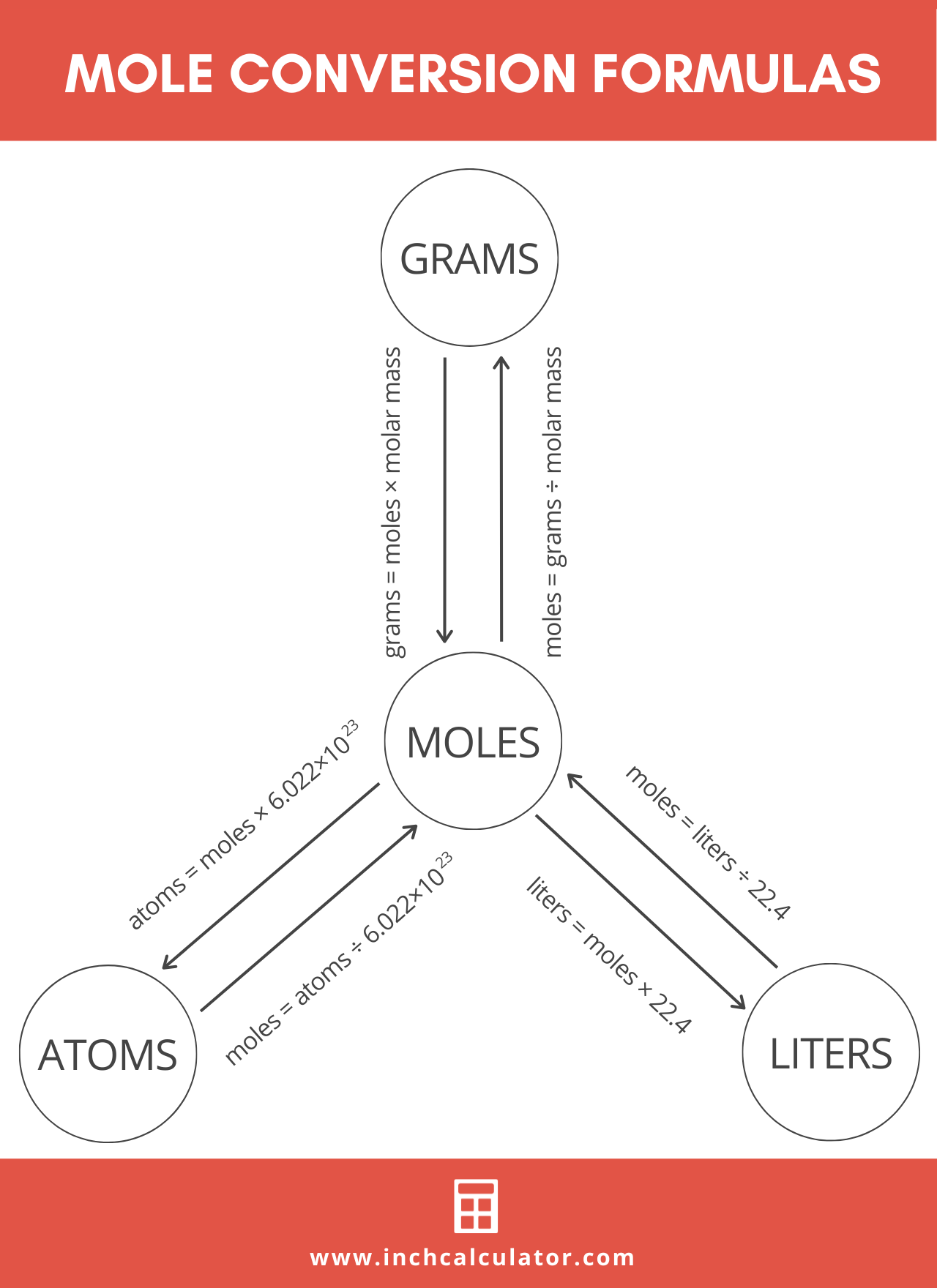
molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises